EPJ D Highlight - Improving tumour radiation therapy: when basic ions break DNA down
- Details
- Published on 30 June 2014
A new study relevant for cancer radiation therapy shows that DNA building blocks are susceptible to fragmentation on contact with the full range of ions from alkaline element species
Scientists now have a better understanding of how short DNA strands decompose in microseconds. A European team found new fragmentation pathways that occur universally when DNA strands are exposed to metal ions from a family of alkaline and alkaline earth elements. These ions tend to replace protons in the DNA backbone and at the same time induce a reactive conformation leading more readily to fragmentation. These finding have been published by Andreas Piekarczyk, from the University of Iceland, and colleagues in a study in EPJ D. They could contribute to optimising cancerous tumour therapy through a greater understanding of how radiation and its by-products, reactive intermediate particles, interact with complex DNA structures.
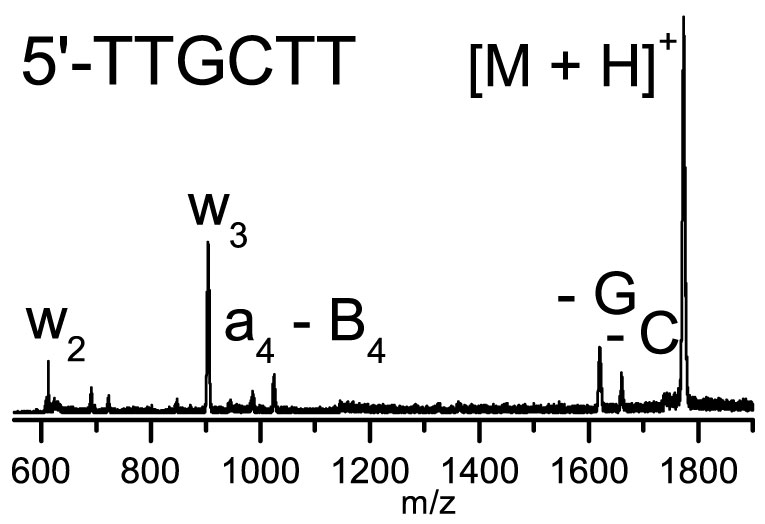
In cancer radiation therapy, it is not the radiation itself that directly damages the DNA strands, or oligonucleotides. But rather, it is the secondary reactive particles, leading to the creation of charged intermediates. Here, the authors have studied one of these charged intermediates in the form of so-called protonated metastable DNA hexamers.
To do so, the authors created selected oligonucleotide-metal-ion complexes that they selected to have between zero and six metal ions. They then followed these complexes’ fragmentation reactions using a technique called time-of-flight mass spectrometry. By comparing the different species, they could deduce how the underlying metal-ion-induced oligonucleotide fragmentation works.
They discovered that metal ion-induced fragmentation of oligonucleotides is universal with all alkaline and alkaline earth metal ions, such as, for example, lithium, Li+; potassium, K+; rubidium, Rb+; magnesium, Mg2+ and calcium, Ca2+. They had previously reached the same conclusion for sodium ions—which are ubiquitous in nature, in the form of sodium chloride, or salt. Once the number of sodium ions per nucleotide is high enough, the study shows, it triggers an unexpected oligonucleotide fragmentation reaction.
A. Piekarczyk, I. Bald, H. D. Flosadottir, B. Ómarsson, A. Lafosse, O. Ingolfssson (2014), Influence of metal ion complexation on the metastable fragmentation of DNA oligohexamers, European Physical Journal D, DOI: 10.1140/epjd/e2014-40838-7





